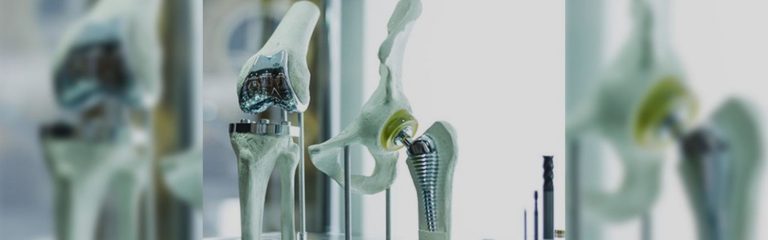
May 18, 2015 – The FDA issued a Class 1 Device Recall Zimmer M/L Taper Hip Prosthesis with Kinectiv Technology.
Due to high levels of manufacturing residue left on the implant components patients are at risk for:
- Allergic Reactions – metal poisoning due to debris and microscopic metal particles in the bloodstream
- Pain and Inflammation – redness, tenderness, swelling in the hip joint,
- Infections – the bone and tissues surrounding the implant
- Loosening and Dislocation – when the implant wears against the bone surfaces, some people have experienced loosening of the replacement and joint dislocation that led to revision surgeries
- Bone Fractures – this can occur in the hip bones due to defects in the neck and stem components
- Bone and Tissue Loss – if a patient has a revision surgery they may suffer bone and tissue loss in the joint.
The FDA has stated that revision surgery to replace defective implants may be necessary.