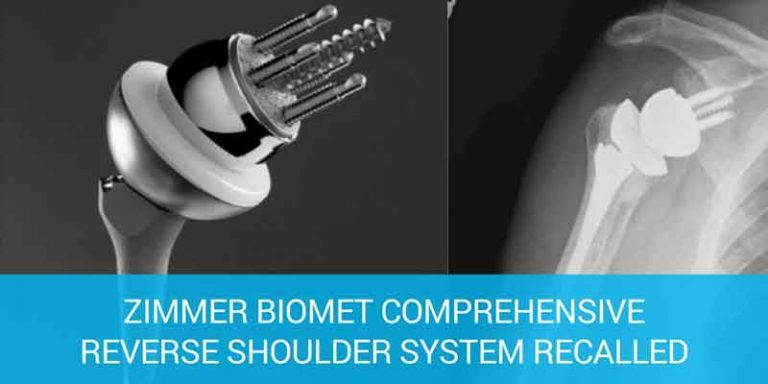
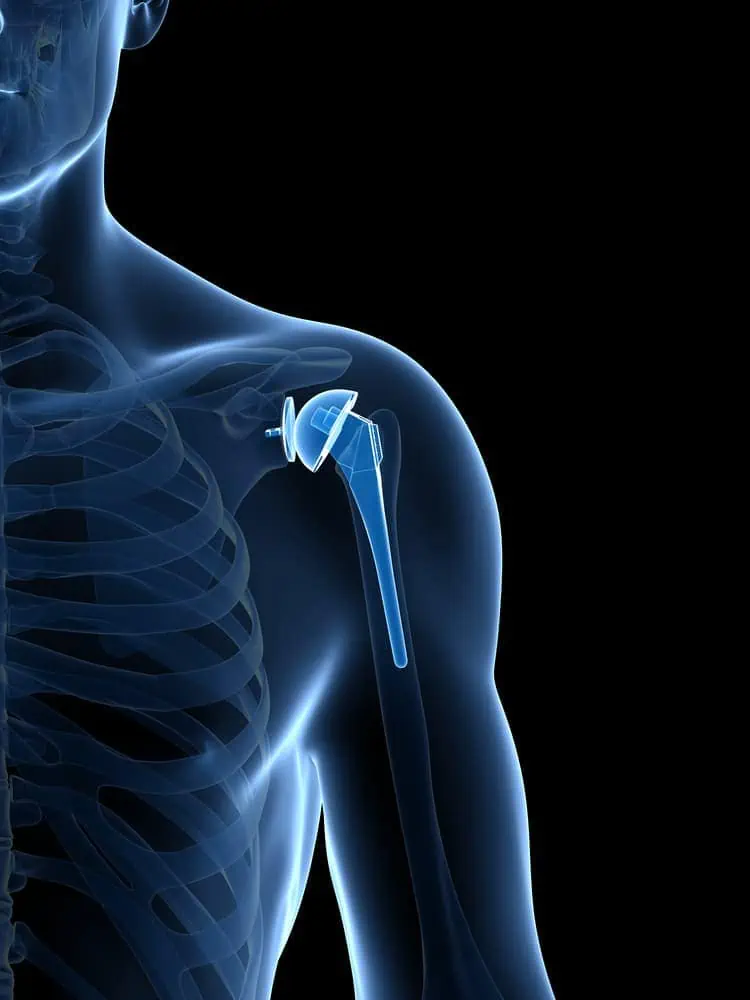
In shoulder replacement surgery, doctors replace the ends of the damaged upper arm bone (humerus) and usually the shoulder bone (scapula) or cap them with artificial surfaces lined with plastic or metal and plastic.
Reverse shoulder replacement surgery is a newer procedure used on people who have painful arthritis in their shoulder and also have damage to the muscles around the shoulder.
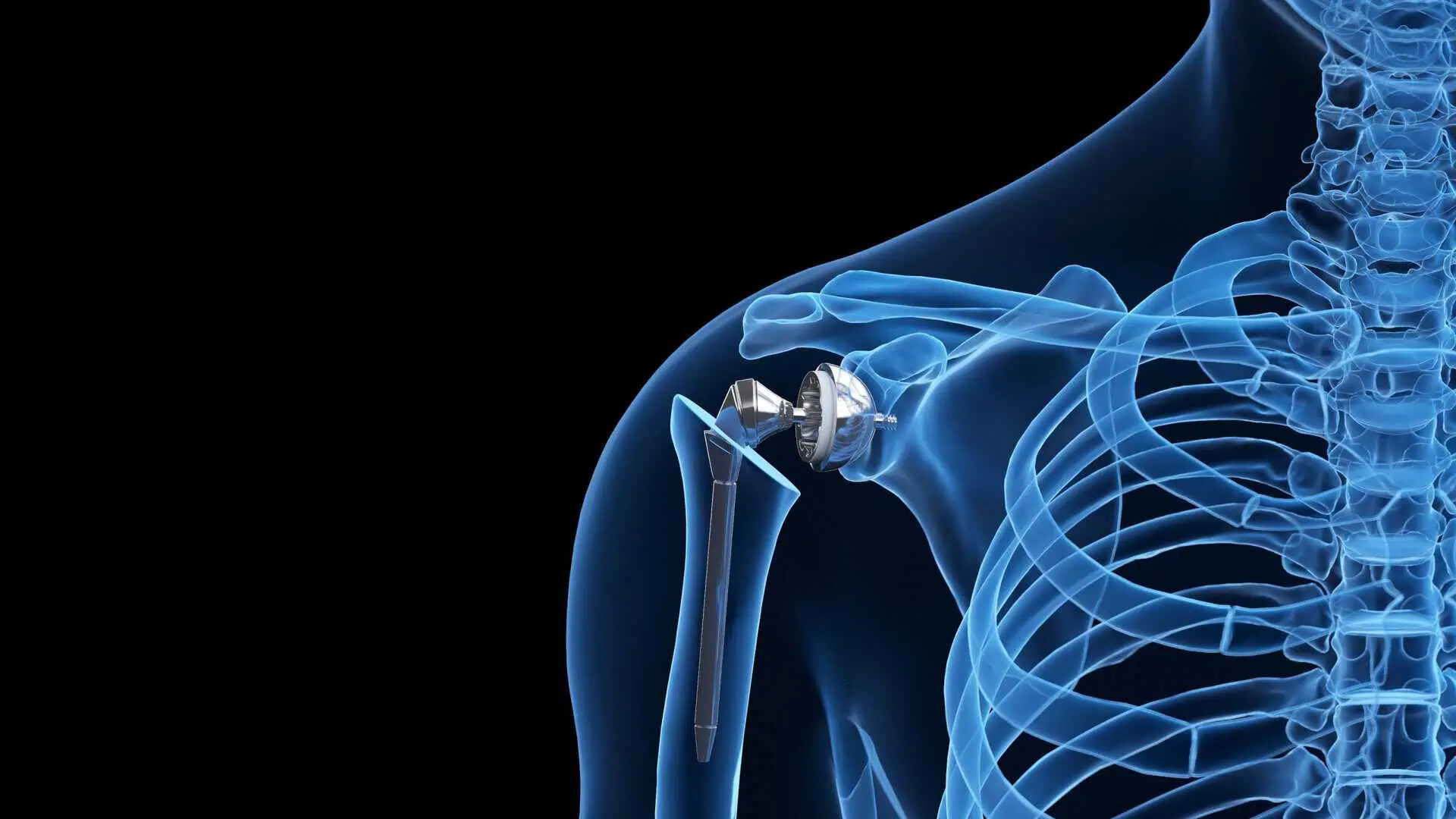
In a reverse shoulder replacement surgery, the surgeon removes the damaged bone and smooths the ends, he or she attaches the rounded joint piece to the shoulder bone and uses the cup-shaped piece to replace the top of the upper arm bone.